What is a patent?
Although rights may vary from country to country, in general, a patent gives the holder the right to exclude others from making, using, selling, offering to sell, and importing the patented invention. Because a patent a “right to exclude,” a patent does not necessarily provide the holder any affirmative right to practice a technology; that practice may fall under a broader patent(s) owned by others.
What is the United States Patent and Trademark Office (PTO)?
The PTO is the federal agency, organized under the Department of Commerce, that administers patents on behalf of the U.S. government. The PTO employs patent examiners skilled in all technical fields in order to appraise patent applications. The PTO also issues federal trademark registrations.
What type of subject matter can be patented?
Patentable subject matter includes processes, machines, compositions of matter, articles, some computer programs, and methods (including methods of making compositions, methods of making articles, and even methods of performing business). The type of inventions that are patentable vary from country to country.
Can software be patented?
Some software is patentable, and OTC evaluates the possibility of filing a patent application on software reported to our office; but in most cases patenting is not the preferred pathway for IP protection for software. This is due to a variety of factors, including the recently heightened burden in proving that a software invention is patentable, challenges in proving infringement if a patent is obtained, and the speed at which software markets and products change. In most cases, copyright protection is sufficient to enable commercialization of software.
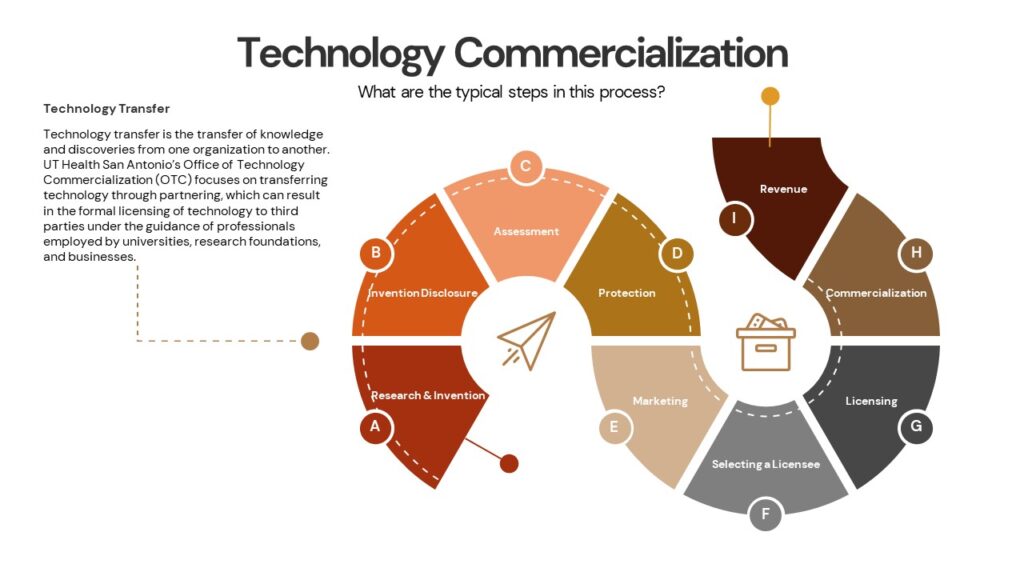
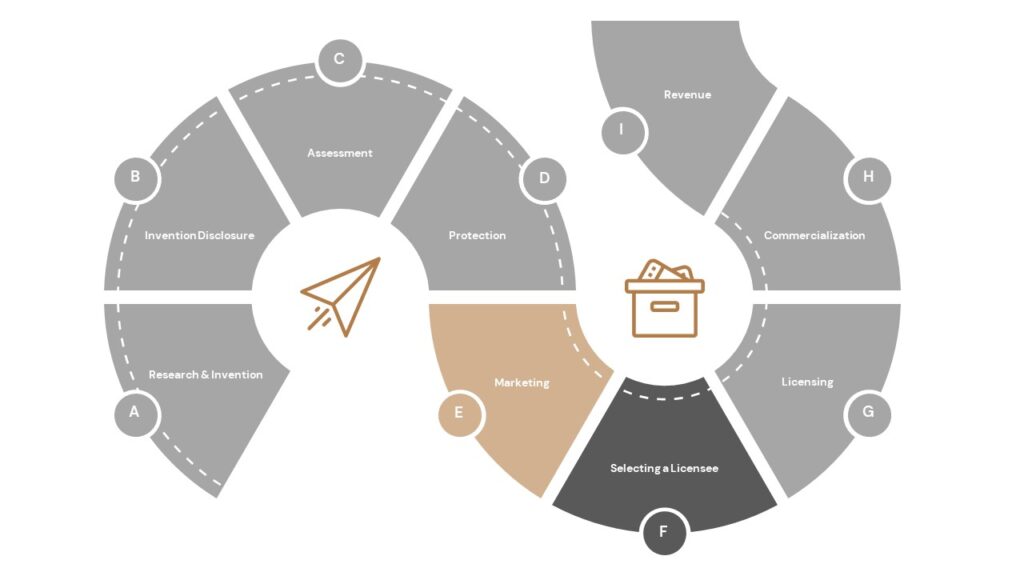
What is the patenting process?
Patent applications are drafted and filed by a patent attorney or a patent agent (a non-attorney with a science education registered to practice before the PTO); OTC, from an approved UT System list of legal firms, retains outside patent counsel to draft and file provisional patent applications in collaboration with the inventor. Our office will most often file a provisional patent application first. This provisional application is not formally reviewed on the merits, but it can serve as a placeholder to establish filing dates that are critical in the patent process. Over the course of the next several years after the initial provisional filing, there may be several additional applications filed both in the US and abroad. While the applications are pending, they will be reviewed by patent examiners at the PTO (or a foreign country’s equivalent patent office). If the examiner has any concerns about the patentability of the technology, the examiner will issue a formal review document called an office action. The office action will describe in detail the issues the examiner has raised, and it will provide a deadline by which our attorneys must file a formal response. We may receive more than one office action during the course of the patenting process. If we are able to overcome each matter raised by the examiner, we will receive a Notice of Allowance. Once we respond to the Notice of Allowance and all other criteria have been met, the PTO will issue the patent.
During the patent process, OTC will continually assess the technology, the market, and its commercialization potential to determine if we should continue to move forward with seeking patent protection. Inventor input is critical at every step of the process. Inventors not only provide most of the detail necessary for the attorney to draft a patent application, but inventors can also provide critical information that may be helpful in overcoming an examiner’s objection.
How are inventors involved in the patenting process?
The patent attorney will ask you to provide as much detailed information as possible, including a Word version of any draft manuscripts or grants, if available. The attorney will also request that you review an application before it is filed. At the time an application is filed, the patent attorney or OTC will ask the inventor(s) to sign a “Declaration” (which basically says that the inventor made the invention and agrees to tell the truth during patent prosecution) and an “Assignment” (which evidences the inventor’s duty to assign the patent to the Board of Regentsof the University of Texas System (“Board”)). In addition, Inventors have an obligation to provide to our patent attorney any relevant publications or information that may be relevant to the invention; failure to disclose such information may render any issued patent unenforceable or invalid.
What does it cost to file for and obtain a patent?
Obtaining a patent is expensive. Filing a U.S. utility patent application typically costs between $10,000 and $15,000. To obtain an issued patent may require an additional $10,000 to $20,000 for patent prosecution. Filing and obtaining issued patents in other countries may cost $20,000 or more per country. Also, once a patent is issued in the U.S or in foreign countries, certain government fees are required to keep the patent alive (referred to as “maintenance fees” in the U.S. and “annuities” in other countries).
What does “first-to-file” patent system mean?
The U.S. changed to a “first-to-file” patent law in 2013. First-to-file is a shorthanded way of referring to how one defines “prior art.” If two inventors independently seek patent protection for the same invention, the inventor that first filed her/his application will be awarded the patent on that invention (assuming that the application fully describes and enables the invention). Under our old system, in many such cases, a process would be used to determine which inventor made the invention first. While there is a lot of literature on the subject, the reality is that in practice our new law will not affect a lot of competing inventors.
What is the effect on patenting if I publish the results of my research?
Publishing or presenting can prevent us from obtaining patent protection, so OTC would like to know about your invention before you publish on it. There are significant differences between the U.S. and other countries as to how early publication affects a potential patent. Once publicly disclosed (published or presented in some form), an invention may have a restricted or minimal potential for patent protection outside of the United States. Since patent rights are affected by publishing or presenting, it is best to submit an invention report or call your licensing specialist at least 3 months before communicating or disclosing your invention to others.
Even after you submit an Invention Disclosure, be sure to inform the OTC licensing specialist assigned to you of any imminent or prior presentation, lecture, poster, abstract, website description, research proposal submission, dissertation/masters thesis, publication, or other public presentation describing any part of the invention. Submitting a manuscript to your licensing specialist prior to submitting to a journal for publication can be extremely helpful for both the marketing and licensing process as well as for the patent attorney assigned to the case.
Is there any effect or implication from the order of inventors named in a patent application or patent?
Not at all. While authorship order in journal publications is relevant to input and perhaps prestige, the order of the inventors on a patent application conveys no advantage to the inventor or patent application.
Are patent applications kept secret?
The PTO holds patent applications confidential until published by the PTO, 18 months after initial provisional filing. After that time, both the application and the PTO’s prosecution materials related to the application are publicly available.
Why file a provisional patent application as opposed to a regular (“nonprovisional” or “utility”) patent application?
In certain circumstances, U.S. provisional patent applications can provide a tool for preserving patent rights while postponing the cost of negotiating the patent application with the PTO. This occurs because a provisional application is not examined during the year in which it is pending, and claims are not required. A regular U.S. application and related foreign applications must be filed within one year of filing the provisional application in order to receive its early filing date. However, an applicant only receives the benefit of the earlier filing date for material that is adequately described and enabled in the provisional application. As a result, the patent attorney likely will need your assistance when an application is filed as a provisional.
What’s different about foreign patent protection?
Foreign patent protection is subject to the laws of each individual country, although in a general sense the process works much the same as it does in the United States. In foreign countries, however, an inventor will lose any patent rights if he or she publicly discloses the invention prior to filing the patent application. In contrast, the United States has a one-year grace period for public disclosures by an inventor.
Who is responsible for patenting?
OTC contracts with UT System approved outside patent counsel for IP protection, thus assuring access to patent attorneys with expertise in diverse technology areas. Inventors work with the patent counsel in drafting the patent applications and responses to worldwide patent offices. OTC staff will select the outside patent counsel based on their expertise in the technology field.
Is there such a thing as an international patent?
Although an international patent does not exist, an international agreement known as the Patent Cooperation Treaty (PCT) provides a streamlined filing procedure for most industrialized nations. The best way to think about it is that while there are international patent applications, there is no such thing as an international patent. For U.S. applicants, a PCT application is generally filed one year after the corresponding U.S. application (either provisional or regular) has been submitted. The PCT application must later be filed in the national patent office of any country in which the applicant wishes to seek patent protection, generally within 30 months of the earliest claimed filing date. The PCT provides two advantages. First, it delays the need to file costly foreign applications until the 30-month date, often after an applicant has the opportunity to further develop, evaluate and/or market the invention for licensing. Second, the international preliminary examination often allows an applicant to simplify the patent prosecution process by having a single examiner speak to the patentability of the claims, which can save significant costs in prosecuting foreign patent applications. An important international treaty called the Paris Convention permits a patent application filed in a second country (or a PCT application) to claim the benefit of the filing date of an application filed in a first country. However, pursuant to this treaty, these so-called “convention applications” must be filed in foreign countries (or as a PCT) within one year of the first filing date of the U.S. application.
What is the timeline of the patenting process and resulting protection?
Currently, the average U.S. utility patent application is pending for about two years, though inventors in the biotech and computer fields should plan on a longer waiting period before a patent is awarded (issued). Once a patent is issued, it is generally enforceable for 20 years from the initial filing of the application that resulted in the patent, if PTO-mandated maintenance fees are paid. The actual life of the patent may vary based on its lineage and whether the PTO grants any additional time extensions.
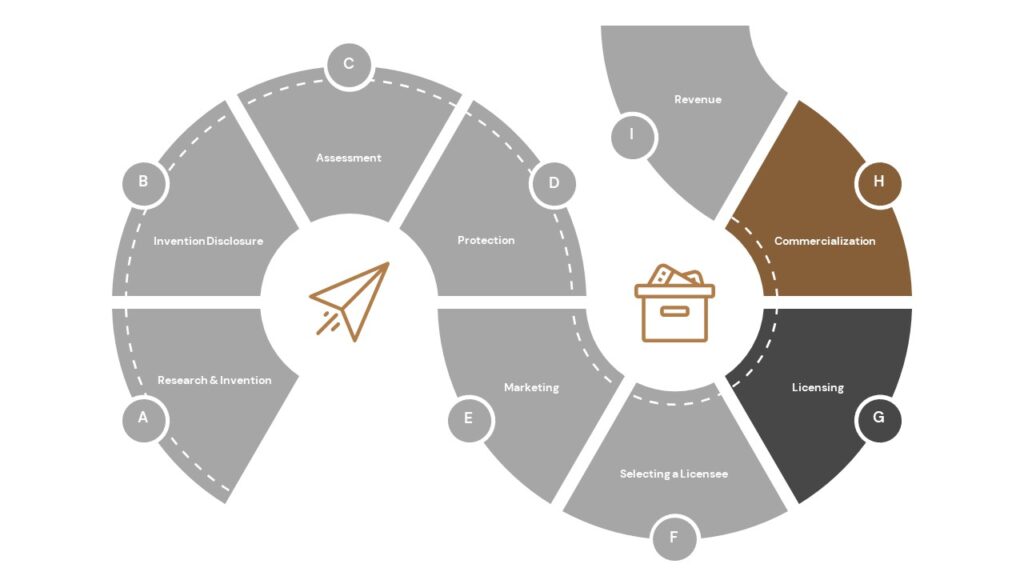
Why does UT Health San Antonio protect some intellectual property through patenting?
Patent protection is often a requirement of a potential commercialization partner (licensee) because it can protect the commercial partner’s often sizable investment required to bring the technology to market. Due to their expense and the length of time required to obtain a patent, patent applications are not possible or appropriate for all UT Health San Antonio intellectual property. We carefully review the commercial potential for an invention before investing in the patent process. However, because the need for commencing a patent filing usually precedes finding a licensee, we look for creative and cost-effective ways to seek early protections for as many promising inventions as possible.
Who decides what gets protected?
OTC utilizes a process that results in a Technology Management Report. The process requires up to 10 weeks to perform before a decision is determined. Numerous factors including the expertise of the inventor(s) are considered. Based on a recommendation from the licensing specialist, the Assistant Vice President for Technology Commercialization ultimately makes the final decision as to whether to file a patent application or seek another form of protection.
Will UT Health San Antonio initiate or continue patenting activity without an identified licensee?
Sometimes. UT Health San Antonio evaluates all technologies on the basis of commercialization potential first. If the invention warrants commercialization, the intellectual property strategy is then considered. OTC bears the expenses of filing a patent application before a licensee has been identified. After UT Health San Antonio’s patent rights have been licensed to a licensee, the licensee generally reimburses OTC for past patent expenses and continues to pay ongoing patent expenses, especially where the license is exclusive. At times UT Health San Antonio must decline further patent prosecution after a reasonable period (often a year or two) of attempting to identify a licensee, or if it is determined that we cannot obtain reasonable claims from the PTO.